Case studies
Explore CMAC’s case studies to see how our research and innovation in medicines manufacturing is delivering real-world impact across industry, regulation, and academia.
ESTAN is a user‑friendly modelling toolbox developed by Loughborough University in collaboration with CMAC. It simplifies complex sensitivity and estimability analyses, helping users build mathematical models and digital twins with stronger predictive performance.
CMAC is levelling up STEM engagement with an immersive VR game that brings digital medicines manufacturing to life for young learners. Developed with The Glasgow School of Art, the experience transforms complex lab science into playful, hands‑on missions that inspire curiosity, build skills, and spark future career ambitions.
CMAC’s Quality by Digital Design (QbDD) approach uses advanced digital tools to rapidly design API manufacturing processes, reducing materials, experimentation, cost and waste while improving efficiency, sustainability and product quality.
CMAC is driving digital transformation in medicines development through Quality by Digital Design (QbDD), a connected approach that brings together data, workflows and automation to make manufacturing more efficient, sustainable and high-quality.
CMAC’s digital‑first process design is cutting pharmaceutical development timelines and material use by an order of magnitude while delivering high‑quality drug substance. By replacing trial‑and‑error experimentation with predictive modelling, it enables faster, more cost‑effective and sustainable manufacturing.
CMAC’s impurity rejection workflow provides industry with a systematic, material‑efficient approach to achieving high‑purity crystalline products in drug manufacturing. Successfully applied by Eli Lilly in clinical development, it enables robust control of impurity‑related CQAs and accelerates crystallisation process design.
Model-based Digitalisation Framework Development for Continuous Manufacturing Processes
Workflows for the Application of a User-friendly Mechanistic Modelling Toolkit
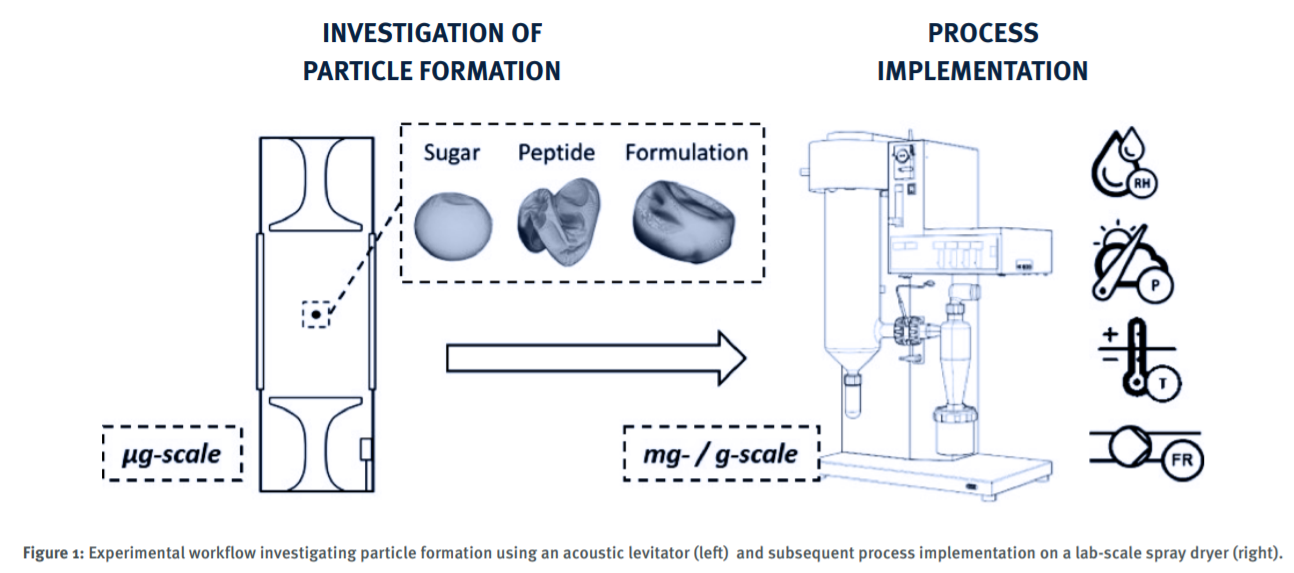
This collaborative CMAC–Eli Lilly project explores spray drying as a faster, scalable alternative to lyophilisation for isolating therapeutic peptides. Through targeted experimentation and advanced data capture, it delivers practical guidance for improving manufacturability and accelerating peptide drug development.
Considerable time has been spent as part of the EPSRC CMAC CIM research campaign, designing and implementing a workflow for continuous cooling crystallisation.
This research explores possibilities to align future value network configurations and disruptive shifts in manufacturing and information technologies.
Computer Aided Process Design (CAPD) and simulation tools have been successfully implemented in the chemical and oil industries since the early 60s to accelerate development and optimise the design and operation of process.
Along with nucleation and growth, agglomeration is a commonly occurring process in crystallisation operations.
The “Make To Order Processing Plants” (MOPPs) project involving CMAC, CPI, AstraZeneca and led by Perceptive Engineering.
The particle engineering research group under the supervision of Prof Jan Sefcik has designed and investigated a novel continuous seed production unit.