Transforming Pharmaceutical Manufacturing with Quality by Digital Design (QbDD)
The CMAC Future Manufacturing Research Hub team used the Quality by Digital Design (QbDD) workflow approach to rapidly design three API manufacturing processes
The challenge
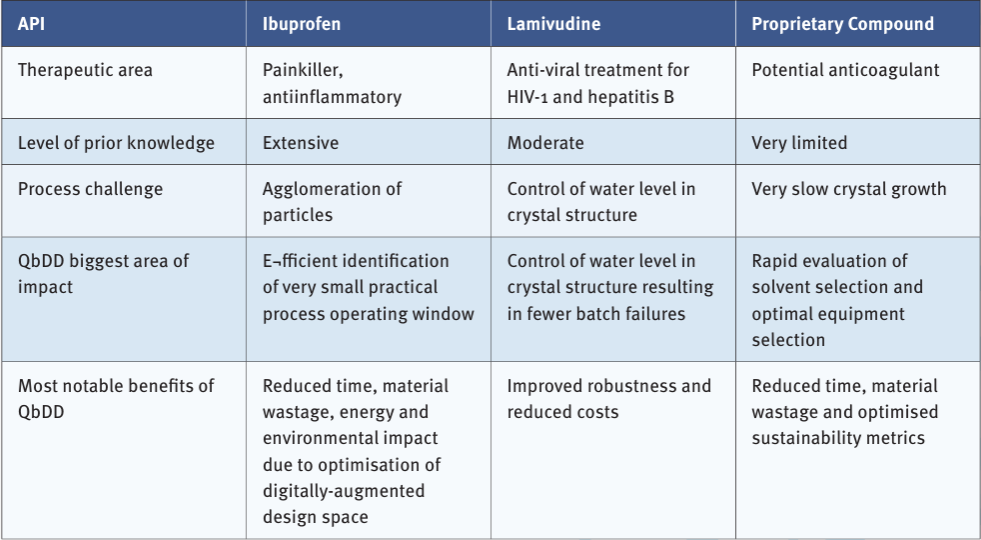
Pharmaceutical manufacturing consists of lengthy, complex processes, with resource-intensive development. There is a need for faster, cheaper, more sustainable practices while maintaining regulatory compliance and ensuring product quality.
Approach
The Quality by Digital Design (QbDD) framework has the potential to transform pharmaceutical manufacturing by integrating advanced digital tools with traditional Quality by Design (QbDD) approaches. In this case study, we demonstrate how a QbDD approach has been used for drug process development of three APIs.
When crystals form they can incorporate solvent molecules into their crystal structure which can affect the chemical stability, ease of formulation, drug release rate, biological effect and cost of manufacture. Lamivudine is such an example when it crystallises in the presence of water (see images). Using a digital model, we were able to predict, and thus control, the amount of water occurring in the lamivudine crystal ensuring we make the desired anhydrous crystal.
Hemi-hydrate (undesired) lamivudine crystals containing water. Images provided by researchers from the EPSRC CMAC Manufacturing Research Hub.
Benefits and expected impact
✔ 65-52% reduction in material usage
✔ 28% reduction in experimentation
✔ Faster development
✔ Cheaper medicines
✔ Greener processes
✔ Less waste
✔ More resilient supply chains
Achieved by: fewer experiments, less materials used, digital multi-objective optimisation and fewer batch failures.
Anhydrous (desired) lamivudine crystals. Images provided by researchers from the EPSRC CMAC Future Manufacturing Research Hub.
Conclusion
QbDD represents a significant advancement in pharmaceutical manufacturing, offering a more efficient, sustainable, and cost effective approach. By combining digital tools with reduced, optimised experimentation, we have shown that QbDD improves and accelerates development pathways, benefitting manufacturers, regulators and patients alike.
Published work
For more information on cases showing implementation of QbDD please read our paper Quality by Digital Design: Exempli cation of a Crystallisation and Isolation Workflow.