Virtual Expert for Regulatory Assistance
Overview

The Virtual Expert for Regulatory Assistance (VERA) is an AI-enabled support tool developed by the Digital CMC CERSI to accelerate the safe and consistent adoption of digital and AI technologies in pharmaceutical CMC.
VERA supports the CERSI mission to advance regulatory science by providing a practical bridge between complex global guidance and real-world implementation. Aligned to the Digital CMC Regulatory Lifecycle (DCRL) framework, it helps users navigate regulatory expectations, identify relevant evidence requirements and support regulator-ready decision-making.
VERA provides a text-based, conversational interface where users can ask natural language questions relating to regulatory expectations, best practices and technical requirements associated with digital CMC tools.
What VERA supports
VERA acts as an intelligent assistant aligned to the DCRL framework, helping users to:
Find and interpret regulatory expectations in a clear and structured way
Define context of use, risk and evidence requirements for digital tools
Support preparation for regulatory submission or inspection
Promote consistency and shared understanding across organisations and regions
Intelligent filtering and continuous updates
A key feature of VERA is its intelligent filtering system, which enables users to refine searches by:
Region
Organisation
Document type
Year range
This metadata-driven approach helps users identify the most relevant regulatory context for their specific application or use case.
The platform is designed to be continuously updated as new guidance documents, standards and best-practice resources emerge, ensuring that the knowledge base evolves alongside the regulatory landscape.
How VERA works
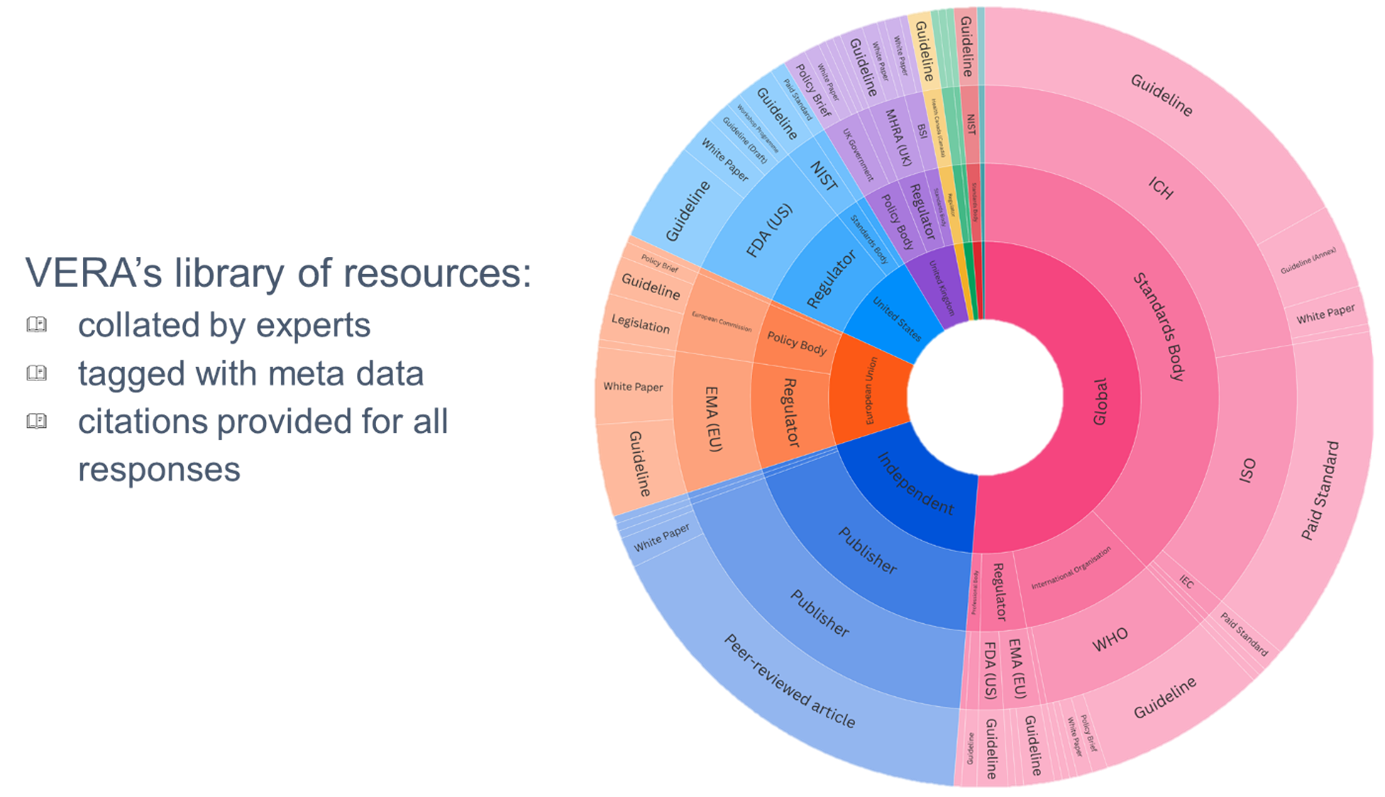
VERA uses an OpenAI large language model (LLM) connected to a curated Retrieval-Augmented Generation (RAG) database containing materials relevant to the Digital CMC CERSI scope.
The knowledge base currently includes more than 300 regulatory guidance documents, standards and scientific best-practice resources, including draft, discussion and published materials, alongside open-access elements of paywalled publications where appropriate.
To improve reliability and reduce hallucination risk, the LLM is constrained to generate responses only from content contained within the RAG database. If information relevant to a query is not available within the knowledge base, VERA will indicate this directly.
All responses include supporting references and relevant source information to improve traceability and transparency.
The VERA Knowledge Base (RAG Database)
At the core of VERA is a curated Retrieval-Augmented Generation (RAG) database, which ensures that responses are grounded in authoritative and up-to-date sources. This database brings together:
Global regulatory guidance (e.g. ICH quality guidelines, FDA, EMA, MHRA and other agency publications)
International standards and frameworks (e.g. ASME model credibility standards, ISO and related best practices)
Scientific literature and peer-reviewed research (covering modelling, digital CMC, AI/ML, and validation approaches)
Industry white papers and cross-sector reports (reflecting real-world implementation challenges and solutions)
CERSI-developed materials (including the DCRL framework, case studies, and practical guidance)
Why VERA matters

This structured evidence base ensures that VERA’s outputs are traceable, transparent and aligned with current regulatory thinking, supporting confidence for both technical experts and non-specialist users. The LLM is restricted to only providing responses from the RAG database and provides the reference(s) and relevant page number(s) when responding to all queries.
VERA exemplifies the Digital CMC CERSI approach: combining science, regulation and innovation to support the responsible adoption of emerging technologies in pharmaceutical CMC.
By making complex regulatory knowledge more accessible, queryable and actionable, VERA helps to:
Accelerate medicines development
Improve global regulatory alignment
Strengthen trust in digital and AI-enabled approaches
Safeguard product quality and patient safety
