Digital Tools in CMC Survey
This study presents findings from a cross-sector industry survey exploring the adoption, use, and regulatory challenges of digital and AI-enabled tools in pharmaceutical Chemistry, Manufacturing and Controls (CMC).
Aims of the study
Assess current adoption of digital and AI-enabled tools across pharmaceutical development and manufacturing
Understand how tools are used (internal decision-making vs regulatory submissions)
Identify key barriers to wider implementation, particularly in regulated environments
Capture industry perspectives on regulatory expectations and future support needs
Inform regulatory science priorities to enable responsible and scalable adoption
Key findings
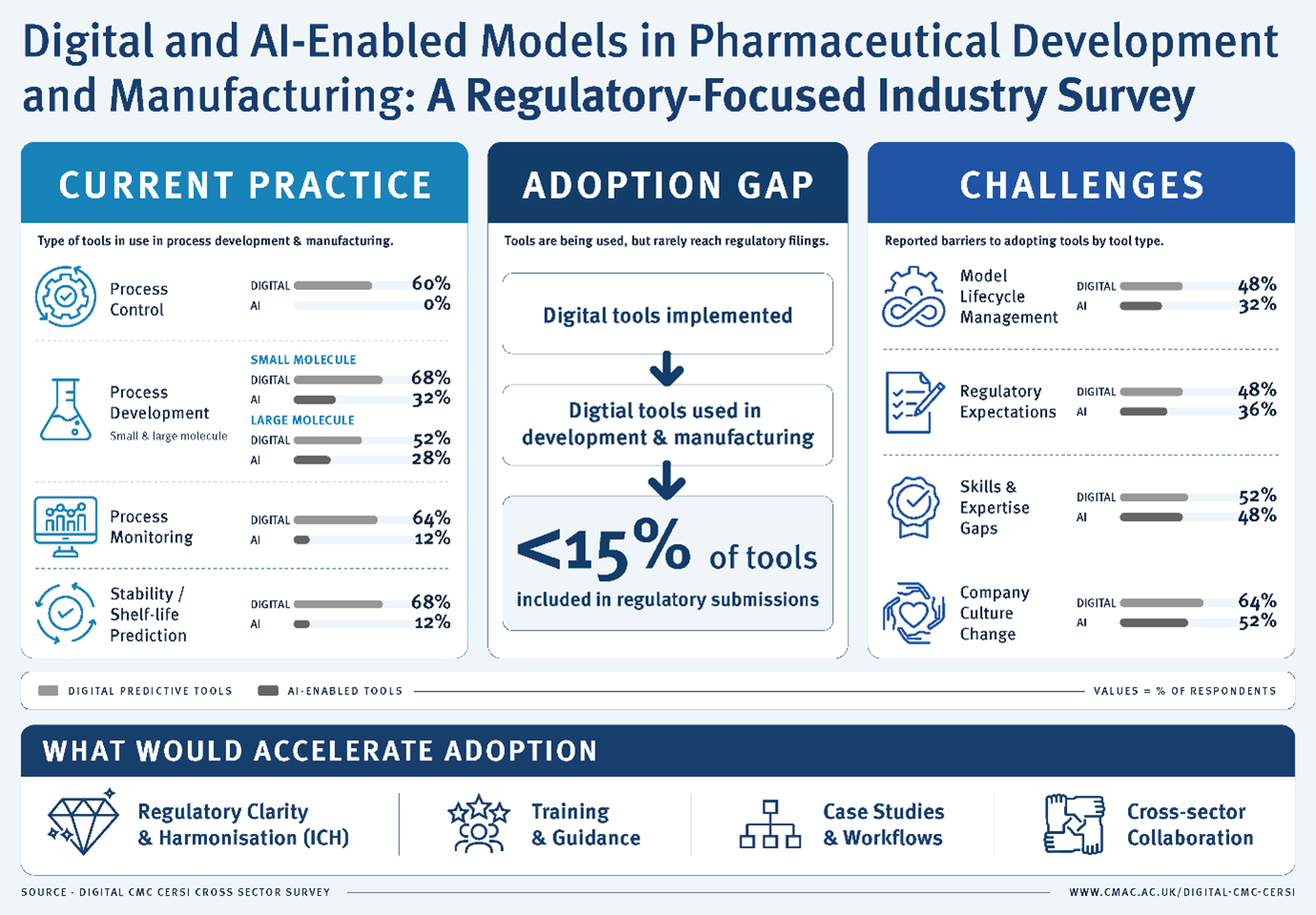
-
Digital tools are commonly used across CMC activities, particularly in process development, monitoring, and control
AI-enabled tools are increasingly adopted, though at lower levels than traditional digital approaches
Adoption is higher in small molecule and drug product applications
-
Fewer than 15% of tools are included in regulatory submissions
Most tools are used internally to support decision-making, rather than as formal regulatory evidence
This highlights a critical “adoption-to-regulation gap”
-
Improved process robustness and product quality
Increased efficiency and reduced development time
Cost savings and accelerated time to market
-
The main challenges are not technical, but systemic:
Uncertainty in regulatory expectations, especially for AI
Skills and expertise gaps in digital and data science
Organisational and cultural barriers to change
Lack of clear frameworks for validation, governance, and lifecycle management
-
Respondents highlighted the need for:
Clear, harmonised international guidance
Regulatory training and upskilling
Case studies and practical workflows
Cross-sector collaboration between industry, academia, and regulators
Implications for Regulatory Science
The findings show that while digital transformation in CMC is advancing, regulatory implementation is lagging.
This creates a need for: Risk-based, proportionate frameworks, clear definitions of context of use, Robust approaches to model credibility and lifecycle management
These are central to the mission of the Digital CMC CERSI in enabling regulator-ready adoption of digital and AI tools.
