The Digital CMC Sandbox
Overview
The Sandbox directly supports the CERSI mission to advance regulatory science by enabling innovation in a structured, risk-informed environment while maintaining product quality, patient safety and regulatory integrity.
In this context, the sandbox allows organisations to test, validate and understand digital tools without regulatory risk, supporting their future use in submissions or inspected environments and helping to align global expectations.
Two Complementary Elements
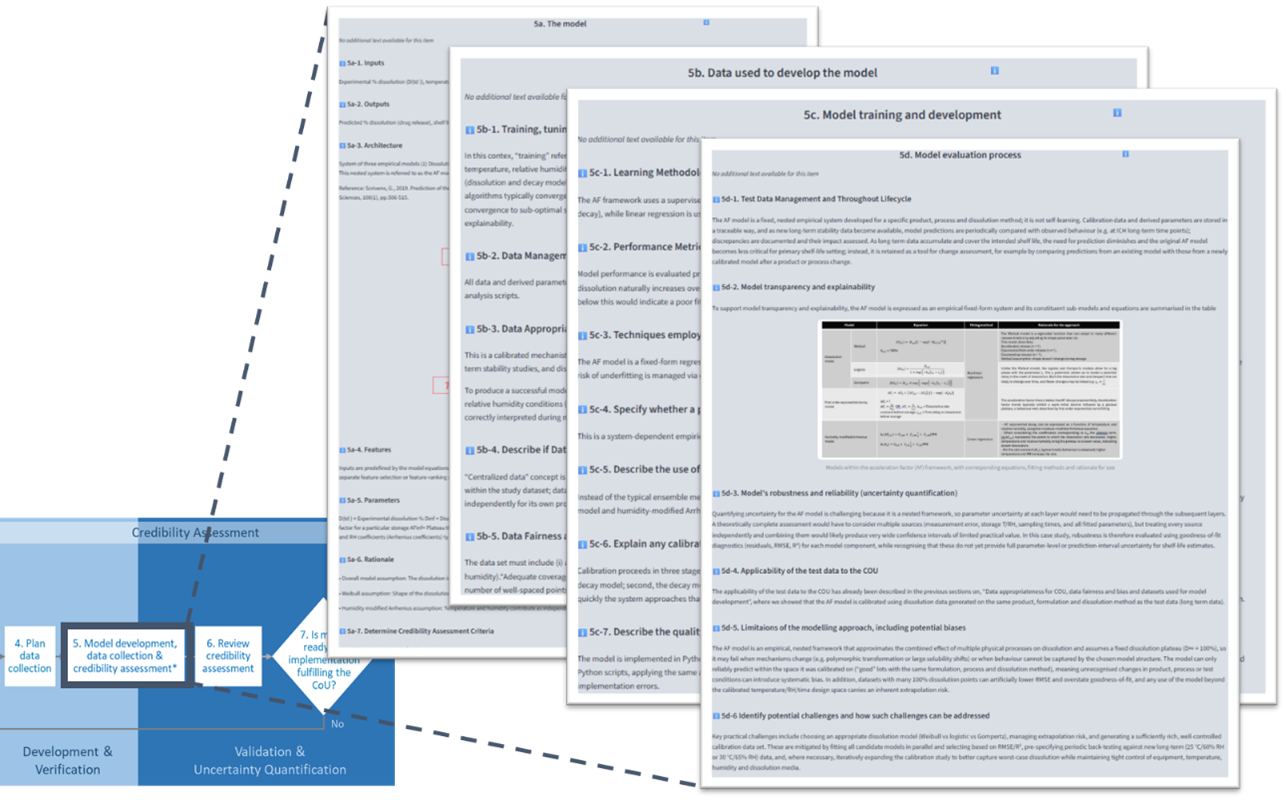
1. Static Environment – Structured Evidence Generation
The static component of the sandbox captures outputs generated through the DCRL workflow. As users progress through each stage, it builds a comprehensive model credibility assessment, including context of use, risk evaluation, data strategy and validation evidence.
Provides a structured, traceable record aligned to regulatory expectations
Supports documentation for regulatory submissions and inspections
Enables consistent interpretation of evidence across organisations and regions
This element ensures that digital tools are supported by robust, transparent and regulator-ready evidence.
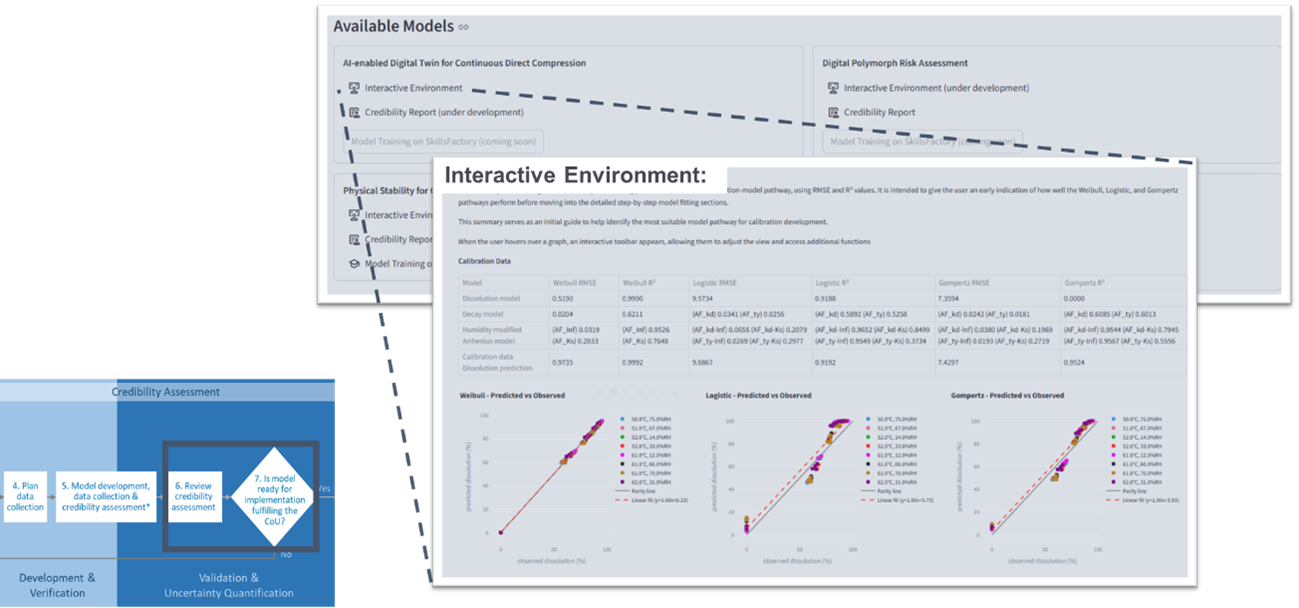
2. Interactive Environment – Dynamic Model Evaluation
The interactive component allows users to actively engage with models in a controlled setting. Users can adjust inputs, rerun models and explore outcomes under different scenarios.
Visualises model performance, uncertainty and robustness
Enables exploration of factors such as data quality, quantity and model assumptions
Supports scenario-based analysis aligned to the defined context of use
This dynamic environment enhances transparency, explainability and trust, helping users understand both the capabilities and limitations of digital tools.
Exemplar Case Studies in the Sandbox
The Digital CMC Sandbox includes four illustrative case studies that demonstrate how the DCRL framework can be applied across different types and regulatory risk levels of computational models. Together, they support the CERSI aim of enabling innovation through practical, regulator-ready examples that build confidence across industry and global regulators.
Case Study 1 – Hybrid (knowledge- and data-driven ML models) Model for Predicting the Solubility of an Organic Compound in a Solvent
Case Study 2 – Data-driven / AI Model: Digital Polymorph Risk Assessment using solid form informatics tools and automated testing
Case Study 3 – Empirical model: Stability Related Dissolution Changes in Oral Solid Dosage Forms to Support Shelf-Life Assignment: An Empirical Modelling Approach
Case Study 4 – Hybrid Digital Modelling Framework for Continuous Direct Compression: Predictive Loss-in-Weight Feeder Models for Soft Sensor Applications
These case studies provide complementary perspectives across model types and use cases, helping users understand how the DCRL framework can be applied consistently. They act as practical reference points to support training, regulatory dialogue, and the safe adoption of digital tools in CMC.

Training and Capability Building
Why it matters
The sandbox also serves as a practical training platform, supporting a wide range of users—from regulators and regulatory professionals to scientists and model developers. Through guided interaction with models and structured credibility assessments, users develop a deeper understanding of:
Model behaviour and uncertainty
Data dependencies and risk considerations
Evidence requirements for regulatory acceptance
More advanced scientific and mathematical training can be layered on where required.
The Digital CMC Sandbox exemplifies the CERSI approach: creating shared environments where innovation and regulation evolve together. By combining structured evidence generation with hands-on evaluation, it helps:
Reduce uncertainty and perceived risk
Build confidence in digital and AI-enabled approaches
Enable consistent, globally aligned regulatory decision-making
Ultimately, the sandbox supports the safe, transparent and effective integration of digital technologies into medicines development and manufacturing.
