Stage 2: Define the Context of Use (CoU)
In accordance with previous ICH, FDA and ISO guidance (ICH Q8(R2), Q12; ISO 22989; FDA, 2025; FDA-EMA, 2026), the Context of Use is defined as a precise description of how the model will be used to address the QoI, including the role of the model in decision-making. This Stage distinguishes between exploratory, supportive, or decision-critical applications and is central to determining the level of regulatory scrutiny.
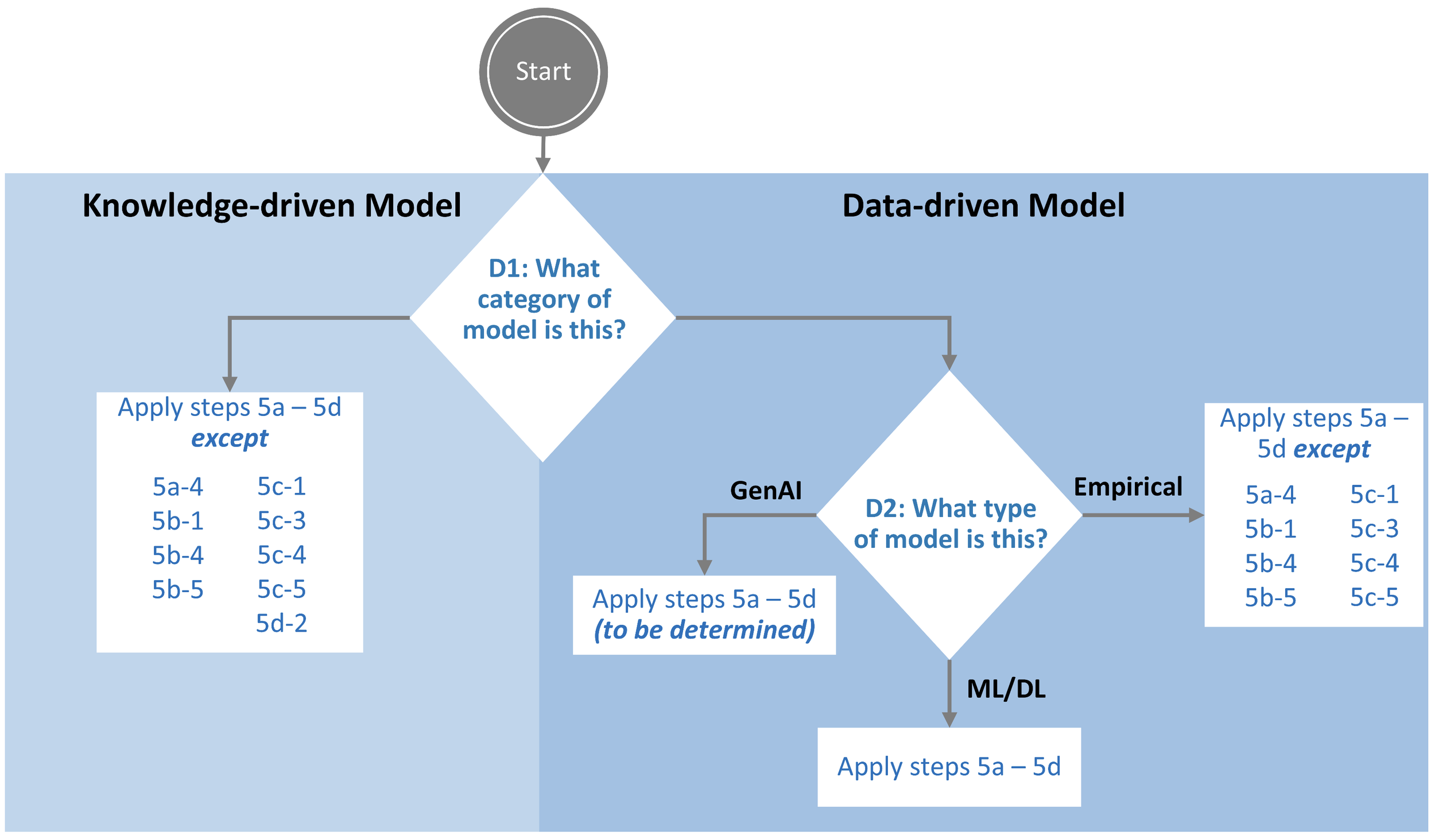
Stage 2a: Model Type Decision
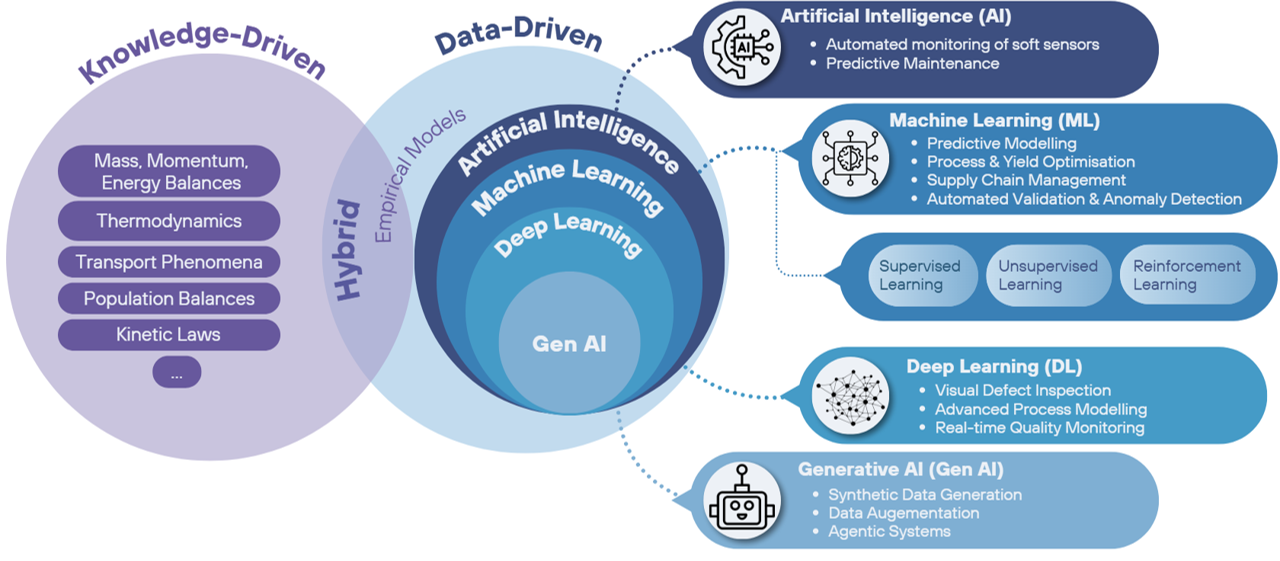
In accordance with guidance from the EMA (EMA, 2024), the model approach should be selected.
Links:
ICH Q8(R2): https://database.ich.org/sites/default/files/Q8%28R2%29%20Guideline.pdf
ICH Q12: https://database.ich.org/sites/default/files/Q12_Guideline_Step4_2019_1119.pdf
ISO 22989: https://www.iso.org/standard/74296.html
FDA-EMA, 2026: https://www.ema.europa.eu/en/news/ema-fda-set-common-principles-ai-medicine-development-0